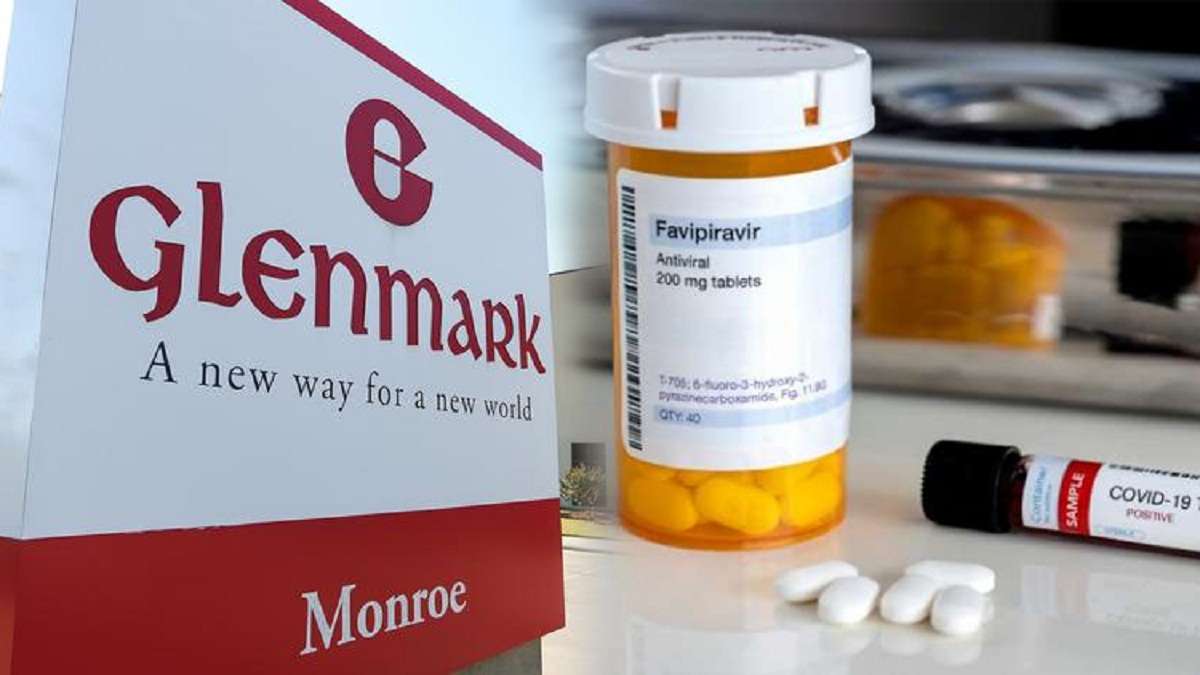
Glenmark medicine for covid ~ It offers rapid reduction in viral load within four days and provides faster symptomatic and radiological improvement. Glenmark Shares Surge Over 35 On Regulatory Approval For COVID-19 Drug Favipiravir being manufactured by Glenmark will be marketed by the name of Fabiflu and will be available in strength of 200. Indeed lately is being hunted by users around us, perhaps one of you personally. People now are accustomed to using the net in gadgets to view video and image information for inspiration, and according to the name of this article I will talk about about Glenmark Medicine For Covid Pharmaceuticals giant Glenmark has launched a COVID drug which has shown promise in treating mild cases.

Fabiflu All You Need To Know About Glenmark S Covid 19 Treatment Medicine Glenmark News India Tv
Source Image @ www.indiatvnews.com
Glenmark medicine for covid | Fabiflu All You Need To Know About Glenmark S Covid 19 Treatment Medicine Glenmark News India Tv
Glenmark medicine for covid ~ Drug firm Glenmark Pharmaceuticals on Saturday said it has launched antiviral drug Favipiravir under the brand name FabiFlu for the treatment of patients with mild to moderate COVID-19 at a price of about Rs 103 per tablet. Glenmark has claimed that the Favipiravir can also be used to good effect in COVID-19 patients with comorbid conditions such as diabetes and heart disease provided the. Glenmark says it is the first company in India to receive approvals for conducting clinical trials of Favipiravir medicine.
Glenmark received the Indian drug regulators approval to manufacture and market a generic of Favipiravir for treating mild to moderate infection. On June 19 2020 Glenmark became the first company in India to receive restricted emergency use approval from Indias drug regulator for Favipiravir FabiFlu making it the first oral Favipiravir-approved medication in India for the treatment of mild to moderate COVID-19. The press release stated On June 19 2020 Glenmark became the first company in India to receive restricted emergency use approval from Indias drug regulator for Favipiravir FabiFlu making it the first oral Favipiravir-approved medication in.
Coronavirus Covid-19 Treatment. FabiFlu the first oral. Priced at Rs 103 per pill it will be availabe as a prescription drug.
In short term but subside as competition increases. The oral antiviral. Favipiravir can be used for coronavirus patients with co-morbid conditions such as diabetes and heart disease with mild to moderate Covid-19 symptoms Glenmark said.
Glenmark to study efficacy of two antiviral drugs for treating Covid-19 Favipiravir is an oral antiviral drug approved in Japan since 2014 for treating novel or re-emerging influenza virus infections. Glenmark will initially enroll 150 patients with mild-to-moderate COVID-19 in a randomized trial that will compare favipiravir with standard supportive care. Glenmark Pharmaceuticals has said it has launched antiviral drug favipiravir under the brand name FabiFlu for the treatment of cases of mild to moderate Covid-19.
Glenmarks PMS study is the first and largest post-marketing study conducted in India on Favipiravir in mild to moderate COVID-19 patients. Favipiravir has demonstrated activity against influenza viruses and has been approved in Japan for Covid-19 treatment Glenmark says. Thirteen sites both Government and private institutions across Mumbai Bangalore Hyderabad Nashik Nagpur and Trivandrum took part.
Glenmark has said the nasal spray is designed to kill the coronavirus in the upper airways. The trial will last for a maximum of. New Delhi India September 15 ANI.
Glenmark concludes post marketing surveillance study on Favipiravir FabiFlu in 1000 COVID patients On June 19 2020 Glenmark became the. Favipiravir can be used for coronavirus patients with co-morbid conditions such as diabetes and heart disease with mild to moderate COVID-19 symptoms Glenmark said. The Glenmark Pharmaceuticals post-marketing surveillance PMS study has shown no safety concerns with the use of.
FabiFlu is the first oral Favipiravir-approved medication for the treatment of COVID-19 the company said in a statement. Glenmark Canadas SaNOtize sign deal for Covid treatment spray in India other Asian countries. Glenmark said in early July 2021 the company presented a proposal to the subject expert committee of CDSCO for emergency approval for import and.
Glenmark Launches COVID-19 Drug Favipiravir At Rs 103 Per Tablet FabiFlu is the first oral Favipiravir-approved medication in India for the treatment of COVID-19 it.
If you are searching for Glenmark Medicine For Covid you've come to the ideal place. We have 14 graphics about glenmark medicine for covid adding images, photos, photographs, wallpapers, and much more. In these webpage, we also have variety of images out there. Such as png, jpg, animated gifs, pic art, logo, blackandwhite, transparent, etc.

Much More Economical Effective Glenmark Defends Price Of Covid 19 Drug Fabiflu After Dcgi Rap
Source Image @ www.news18.com










0 comments:
Post a Comment